Research ethics is concerned with the standards of conduct for scientific researchers (WHO, n.d.). It is a specialized discipline that studies the norms for conduct that distinguish between acceptable and unacceptable behavior when conducting research (Resnik, 2020). Research ethics are important because following the set ethical norms promotes the aims of research such as knowledge, truth, and avoidance of error. Some examples of ethical standards in research would be prohibitions against fabricating, falsifying, or misrepresenting research data.
In research, it is crucial to adhere to ethical principles to protect the welfare, rights, and dignity of the research participants. This is also why it is important for researches involving human subjects to pass through the Research Ethics Committee to undergo review to ensure that ethical standards are strictly implemented and followed.
Adhering to research ethics is also important in non-health disciplines. For instance, social science research, which is concerned with complex issues such as cultural, legal, economic, and political phenomena (Freed‐Taylor, 1994, as cited in Mollet, 2011), must ensure that the research process, along with its findings, are both valid and trustworthy (Mollet, 2011). There are also several ethical issues within social science and humanities research such as those research conducted without consent, not safeguarding the confidentiality and anonymity of participants, fraud, and conducting research with more risks than benefits. These ethical issues become crucial elements in such research. Similar to health-related disciplines, it is also mandatory for social researchers to apply for ethical clearance when dealing with research involving human subjects. In sociological research, the majority of researcher works rely on data involving humans, such as interviews with actively cooperating subjects, records relating to persons still living or recently alive, unobtrusive observation of live actors, or participant studies within interacting groups, and only a few sociologists use historical documents or cultural products as data. Research in sociology also typically focuses on large study populations. Thus, research ethics in this field is of great importance because the process and application of sociological inquiry may significantly affect large numbers of subjects in an unfavorable manner (Encyclopedia of Sociology, n.d.). According to the European Commission (2018), recent technological development and political upheavals raised new research ethics concerns, which further demands the strict implementation of research ethics to identify ethically problematic areas in social science research.
Ultimately, in all disciplines, the ethical principles involving human subjects should be conducted in accordance with the following principles: people must be respected, benefits must be maximized, harms should be minimized, harm shall not be done, and subjects in studies must be treated equally (SBREC Flinders University, 2007, as cited in Mollet, 2011).
References:
“Ethics in Social Research .” Encyclopedia of Sociology. . Retrieved April 25, 2022 from Encyclopedia.com: https://www.encyclopedia.com/social-sciences/encyclopedias-almanacs-transcripts-and-maps/ethics-social-research
European Commission. (2018). Ethics in Social Science and Humanities. Retrieved from https://core.ac.uk/download/pdf/156620447.pdf
Mollet, J. (2011). Ethical Issues in Social Science Research in Developing Countries: Useful or Symbolic. In R. Cribb (Ed.), Transmission of academic values in Asian Studies: workshop proceedings. Canberra: Australia-Netherlands Research Collaboration
Republic Act No. 10532. (2013). Retrieved 4 May 2022, from https://lawphil.net/statutes/repacts/ra2013/ra_10532_2013.html
Resnik, D. (2020). What Is Ethics in Research & Why Is It Important? Retrieved from https://www.niehs.nih.gov/research/resources/bioethics/whatis/index.cfm
World Health Organization. (n.d.). Ensuring ethical standards and procedures for research with human beings. Retrieved from https://www.who.int/activities/ensuring-ethical-standards-and-procedures-for-research-with-human-beings
Republic Act No. 10532
The Philippine National Health Research System Act of 2013, also known as Republic Act No. 10532, is an act institutionalizing the Philippine National Health Research System. It aims to improve Filipinos’ health status, productivity, and quality of life by ensuring that health research is linked to the health system needs, among others. Republic Act 10532 also mentions the importance of the Philippine Health Research Ethics Board or PHREB in ensuring adherence to the universal principles for the protection of human participants in research. As stated in Section 12, the PHREB was created under DOST Special Order No. 091 s. 2006 sets the ethical standards and guidelines for research regarding human health, and it also provides the guidelines for ethics review committees and research ethics review. The functions of the PHREB also include:
- monitoring and evaluating the performance of institutional ethics review committees in accordance with procedures outlined in a prior agreement,
- promoting the establishment of functional and effective ethics review committees,
- providing advice, and giving recommendations to the PNHRS Governing Council and other appropriate entities regarding programs, policies, and regulations as they relate to ethical issues in human health research, and
- contributing to discourses and discussions on ethical issues in human health research, among others.
Link: https://www.officialgazette.gov.ph/2013/05/07/republic-act-no-10532/
Memorandum No. OVPAA 2020-145
The Office of the Vice President for Academic Affairs issued a directive on the Ethics Review of Research Projects Involving Human Subjects on November 30, 2020. This is the official statement from the UP System reminding the UP community of the need for ethics review on research projects, particularly those involving human participants, and forming a PHREB-accredited Research Ethics Committee in each constituent university.
The UP Open University Institutional Research Ethics Committee (UPOU IREC) is the body that will implement the university policy of overseeing the ethical soundness of the institutional research produced, including any research conducted by faculty members, students, REPS, and staff. All institutional research proposals must undergo ethics review and shall gain certification clearance from the UPOU IREC.
The UPOU IREC serves the following functions:
- To develop, recommend, and implement guidelines on the ethical review of research proposals;
- To ensure satisfactory review of submitted research proposals by following standard operating procedures;
- To issue ethical clearance to approved research proposals;
- To ensure the protection of the rights and the well-being and safety of human participants in research;
- To ensure the protection of the integrity of the scientific data; and,
- To address and act on concerns of research participants and other stakeholders.
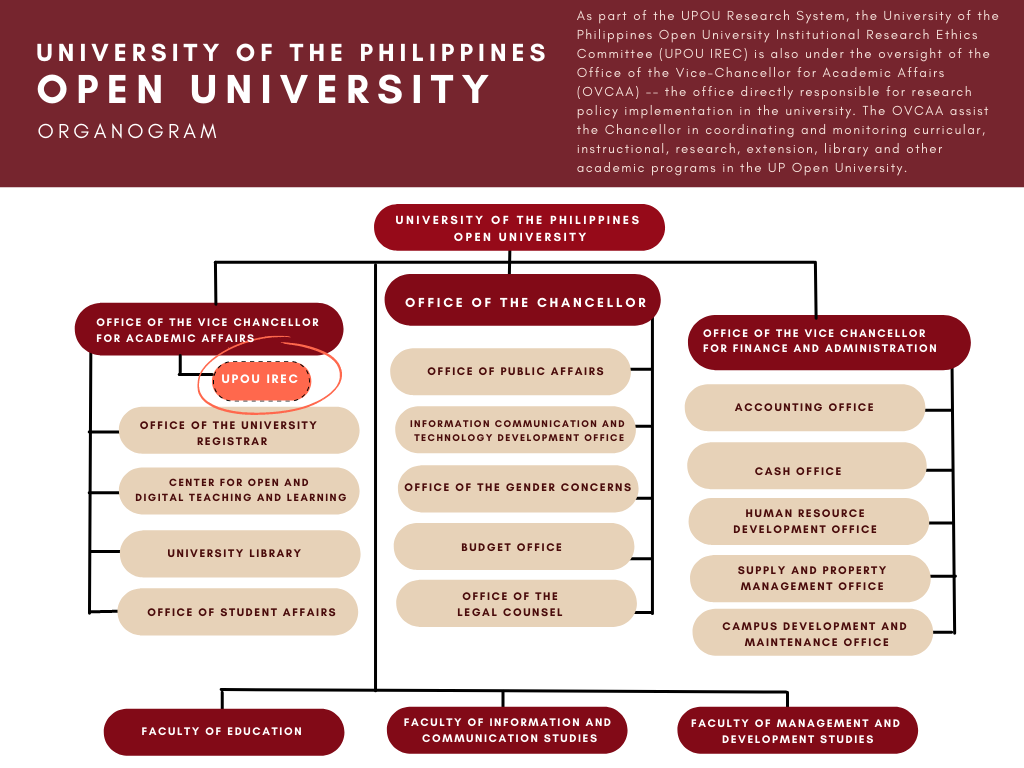
The IREC is part of the UPOU Research System, which is under the oversight of the UPOU Chancellor. The University commits to the establishment and maintenance of an effective ethics review system with the following objectives:
- Establish and maintain the UPOU Institutional Research Ethics Committee (UPOU IREC), determine its composition, and appoint members to the committee;
- Ensure adequate resources to enable the UPOU IREC and its members to discharge their duties and responsibilities in an effective and timely manner, while providing for annual review of resources
- Ensure independence in review and decision
- Manage conflicts of interest and ensure recording of how an institution resolved or managed the conflict of interest
- Accept legal responsibility for the decisions of the UPOU IREC
- Monitor approved proposal for all researches that are done within its jurisdictional premises
- Promote responsible publication and dissemination of research findings
- Ensure support for training in human subject protection for investigators, Good Clinical Practice (GCP), research methods involving human experimentation, confidentiality, data storage and records keeping, as well as regulation and governance for researchers and staff
- Perform and document quality assurance activities to ensure compliance with international and national accreditation bodies
- Provide guidelines for the preparation of Memorandum of Agreement (MOA), Memorandum of Understanding (MOU), Clinical Trials Agreement (CTA), Material
- Transfer Agreement (MTA), and other agreements for multi-institutional and multi-center studies, regarding ownership of data, storage of data, registries, databases, as well as human samples
- Provide guidelines for management and retention of human samples and research data, as well as their secure and safe disposal
- Provide the policies on the ownership of and access to databases, registries, and archives that are consistent with confidentiality requirements, laws, privacy rules, and other guidelines including funding agreements
- Provide guidelines/policies on ownership of data, database and registry ownership, as well as agreements covering ownership of data whenever researchers or staff leave or transfer to other institutions
As part of the UPOU Research System, the IREC is also under the oversight of the Office of the Vice-Chancellor for Academic Affairs (OVCAA) — the office directly responsible for research policy implementation in the university. The OVCAA assist the Chancellor in coordinating and monitoring curricular, instructional, research, extension, library and other academic programs in the UP Open University.

UPOU IREC’s Consultation Meeting Recording and Files are now available
In conjunction with the implementation of the Memorandum No. OVPAA 2020-145 issued by the UP System in 2020, we, the UPOU Institutional Research Ethics Committee, would like to solicit feedback from stakeholders on the most recent version of the UPOU IREC Standard Operating Procedures in order to improve the clarity, feasibility, and completeness of our policies and procedures.
Check out the recordings here:
Initial Submissions
Post-Clearance Submissions
External Transactions
Internal Transactions